
Place a half-dollar or 10 new pence on the table and on one side of the coin a small cork disk. How can you move the cork to the exact centre of the coin without touching it!
Pour water on to the coin - drop wise, so that it does not spill over - to form a water mountain over the surface. At first the force of gravity holds the cork on the edge of the slightly curved water surface. If you now pour on more water, the pressure of the water on the edge increases, while it remains constant on the top. So the cork moves up the hill to the middle, which is the region of lowest pressure.
Ship!!!
Mountain Of Water
 Fill a dry glass just full with tap water, without any overflowing. Slide coins carefully into the glass, one after the other, and notice how the water curves above the glass.
Fill a dry glass just full with tap water, without any overflowing. Slide coins carefully into the glass, one after the other, and notice how the water curves above the glass.
It is surprising how many coins you can put in without the water spilling over. The water mountain is supported by surface tension, as though it is covered by a fine skin. Finally, you can even shake the contents of a salt cellar slowly into the glass. The salt dissolves without the water pouring out.
Water knots
 An empty two-lb. can is pierced five times just above the lower edge with a thin nail. The first hole should be just over an inch from the fifth. Place the tin under a running tap, and a jet will flow from each hole. If you move your finger over the holes, the jets will join together. The water particles are attracted to one another and produce a force acting into the interior of the liquid, the surface tension. It is also this force which holds a water droplet together. In our experiment the force is particularly clear, and it diverts the jets into a sideways are and knots them.
An empty two-lb. can is pierced five times just above the lower edge with a thin nail. The first hole should be just over an inch from the fifth. Place the tin under a running tap, and a jet will flow from each hole. If you move your finger over the holes, the jets will join together. The water particles are attracted to one another and produce a force acting into the interior of the liquid, the surface tension. It is also this force which holds a water droplet together. In our experiment the force is particularly clear, and it diverts the jets into a sideways are and knots them.
String of pearls

Let a fine jet of water pour on a finger held about two inches under the tap. If you look carefully, you will see a strange wave-like pattern in the water. If you bring your finger closer to the tap, the waves become continuously more ball-shaped, until the water jet resembles a string of pearls. It is so strongly obstructed by the finger that because of its surface tension - the force that holds the water particles together - it separates into round droplets. If you take your finger further away from the tap, the falling speed of the water becomes greater, and the drop formation is less clear.
Ice hook

Who can hook an ice cube from a bowl of water with a match? A trick makes it quite easy: place the match on the ice cube and scatter some salt over it. In no time the match is frozen solid, and you can lift it together with the ice cube from the dish.
Salt water does not freeze as easily as ordinary water, and scattering salt on ice makes it melt. The salt grains on the ice cube also do this. However, when a substance melts, heat is consumed at the same time. This heat is taken from the moisture under the match, where no salt fell, in this case - and it freezes.
Cutting Through Ice
 Place an ice cube on the cork of a bottle. Fix two objects of equal weight on a piece of wire, hang the wire over the ice and place the whole lot out of doors in frosty weather. After a certain time the wire will have cut through the ice without dividing it.
Place an ice cube on the cork of a bottle. Fix two objects of equal weight on a piece of wire, hang the wire over the ice and place the whole lot out of doors in frosty weather. After a certain time the wire will have cut through the ice without dividing it.
This trick of nature is explained by the fact that ice melts when it is subjected to pressure. Water is formed where the wire is resting, while it immediately freezes again above it. Skating is only made possible by slight melting of the ice under the moving surface, which reduces the friction.
Iceberg
 Place a cube of ice in a tumbler and fill it to the brim with water. The ice cube floats and partly projects from the surface. Will the water overflow when the ice cube melts!
Place a cube of ice in a tumbler and fill it to the brim with water. The ice cube floats and partly projects from the surface. Will the water overflow when the ice cube melts!
The water increases its volume by one-eleventh when it freezes. The ice is therefore lighter than water, floats on the water surface and projects above it. It loses its increased volume when it melts and exactly fills the space, which the ice cube took up in the water. Icebergs, which are a danger to navigation, are therefore especially harmful because one only sees their tips above the water.
Column of ice

Place an ink bottle filled to the brim with water in the freezing compartment of a refrigerator. Soon a column of ice will stick up out of the bottle.
Water behaves oddly: when warm water-cools it contracts, but if the temperature falls below 40C, it suddenly begins to expand again. At 0 C it begins to freeze, and in doing so increases its volume by one-eleventh. This is the reason why the ice sticks out of the bottle. If you had closed it, it would have cracked. Think about burst water pipes in winter and frost cracks on roads, in which water collected under the asphalt freezes.
Producing cold

With a rubber band fix a wad of cotton wool over the mercury bulb of a room thermometer. Note the temperature, damp the cotton wool with eau-de-cologne, and whirl the thermometer round on a string for a time. The temperature drops considerably. The alcohol in the eau-de-cologne evaporates quickly and so uses up heat. The draught caused by whirling the thermometer round accelerates the process and the heat consumption rises. In a refrigerator a chemical liquid evaporates in a container. The large amount of heat needed for this is taken from the food compartment.
Where Is The Wind Coming From?
 Moisten your finger and hold it straight up in the air. You will notice at once that one side of the finger is cold. This is the direction from which the wind is coming. Heat is used up when a liquid vaporizes or evaporates. The wind accelerates the evaporation of the moisture on the finger and you will notice even with a weak air current the greater heat loss on the side facing the wind. Anybody who keeps on a wet bathing costume after a swim will shiver even in the heat. The water takes heat from the body as it evaporates.
Moisten your finger and hold it straight up in the air. You will notice at once that one side of the finger is cold. This is the direction from which the wind is coming. Heat is used up when a liquid vaporizes or evaporates. The wind accelerates the evaporation of the moisture on the finger and you will notice even with a weak air current the greater heat loss on the side facing the wind. Anybody who keeps on a wet bathing costume after a swim will shiver even in the heat. The water takes heat from the body as it evaporates.
Steam boat

Break off the head of a match and drop some glue on to the end. If you place the match in a dish of water it moves jerkily forward.
The glue contains a solvent, which evaporates to give a vapour. It puffs out from the drop in invisible little clouds, giving the match a small push each time.
Eventually so much of the solvent has escaped that the glue becomes solid. In a dried drop of glue you can still see the residual solvent vapour as small bubbles.
Bath game with a coin

Stretch a strip of cellophane (not plastic foil), 1 inch wide, tightly over a soup plate and fasten the ends with adhesive tape. Lay on the middle of the strip an average-sized coin and pour water into the dish up to about 3-inch under the coin.
The coin sinks slowly and reaches the water after several minutes.
The water vaporises, the cellophane absorbs the water particles from the air and expands until it reaches the water. But strangely enough it soon begins to tighten again, and the coin rises again slowly to its original position.
Water from the Desert
 We still read in the newspapers of people dying of thirst in the desert, but many of them could help themselves in this emergency. An experiment on a small scale in a sandbox will show you how to do it. Dig a fairly deep hole and place a beaker in the middle. Spread a suitably sized piece of transparent plastic foil over the edge of the hole and lay a small stone in its centre so that it dips down to the beaker in the shape of a funnel. The edges are fixed firmly into the sand. Soon, especially in sunshine, small drops of water form on the underside of the foil. They become larger and larger and finally flow into the beaker. The effect of the sun is to heat the ground strongly under the foil. The moisture held in the sand evaporates until the enclosed air is so saturated that small drops of water are deposited on the cooler foil. Even desert sand contains some moisture. If you also place cut up cactus plants into the hole, you will obtain enough water to survive.
We still read in the newspapers of people dying of thirst in the desert, but many of them could help themselves in this emergency. An experiment on a small scale in a sandbox will show you how to do it. Dig a fairly deep hole and place a beaker in the middle. Spread a suitably sized piece of transparent plastic foil over the edge of the hole and lay a small stone in its centre so that it dips down to the beaker in the shape of a funnel. The edges are fixed firmly into the sand. Soon, especially in sunshine, small drops of water form on the underside of the foil. They become larger and larger and finally flow into the beaker. The effect of the sun is to heat the ground strongly under the foil. The moisture held in the sand evaporates until the enclosed air is so saturated that small drops of water are deposited on the cooler foil. Even desert sand contains some moisture. If you also place cut up cactus plants into the hole, you will obtain enough water to survive.
Hygrometer

Coat a strip of writing paper two inches long with glue and roll it onto a sewing needle. Stick a strip of shiny photographic paper about 3 inch wide and one foot long onto its end so that its shiny surface faces the glue-covered side of the writing paper. The filmstrip is rolled round the needle like a clock spring. Punch a small hole through the middle of the bottom and lid of a furniture polish tin, and also air holes in the bottom. File off the metal projections formed. Push the needle through the central holes and stick the end of the filmstrip firmly to the side of the tin.
Fix a paper pointer in front of the needle with a cork disk, and a bead behind it. The gelatin layer of the photographic film expands - in contrast to the paper layer - with increased air humidity, causing it to wind up sharply, and move the pointer to the right. When the humidity of the air falls, the pointer returns to the left.
Weather station

Fix a dry pinecone on to a small piece of wood with sealing wax or glue. Stick a pin into one of the central scales and place a straw over it. Put the cone out-of- doors, protected from the rain. The straw moves according to the state of the weather. Fix up a scale.
This simple hygrometer was built by nature. The pinecone closes when it is going to rain, to protect the seeds from damp. The outside of the scales absorbs the moisture in the air, swells up and bends - a process which you can also observe with a piece of paper which is wet on one side.
Rain In The Room

Rain after sultry days makes the inside of the windowpane suddenly sweat. You can distinguish the tiny water droplets through a magnifying glass. Where do they come from?
After it has been raining the air outside cools sharply because the water evaporates and thus uses heat. The warm air in the room, which is saturated with water vapor, especially from cooking, cools down only slowly on the windowpane. But cold air cannot hold so much moisture as warm air, and therefore loses some of it on to the pane. It forms water droplets - exactly as when it is raining out-of- doors and moist, warm air meets cold air.
Hovercraft

Place a tin lid on a hot plate and heat it well (take care!). If you then let a few drops of water fall on the lid, you will observe a small natural phenomenon. The drops are suspended in the air like hovercraft and whiz hissing to-and-fro for a while.
On contact with the heated metal the water drops begin to evaporate at once on the underside. Since the steam escapes with great pressure, it lifts the drops into the air. So much heat is removed from the drops by the formation of steam that they do not even boil.
Jet boat

Bore a hole from the inside through the screw top of an aluminium pill tube about four inches long, and pour some water into the tube. Fix the tube in an empty sardine can into which you have fixed three candle stumps and place the can in water. If you light the candles the water soon boils, and the jet of steam escaping from the back drives the boat.
Steam is formed in the boat’s boiler when the water boils. Because it expands sharply, it escapes at high pressure through the nozzle and causes a recoil. Do the experiment in calm weather!
Paper saucepan

Do you believe that you can boil water in a paper cup over a naked flame or in the embers of a fire!
Push a knitting needle through the rim of a paper cup containing some water, hang it between two upright bottles and light a candle under the cup.
After a little while the water boils - but the cup is not even scorched.
The water removes the heat transferred to the paper and begins to boil at a temperature of 212F or 100C.
The water does not get any hotter, so the paper does not reach the temperature, which is necessary for it to burn.
Fire Under Water
 Warm the base of a candle stump and stick it in a bowl. Fill the bowl with cold water up to the rim of the candle. If you light the wick it burns until it is under the surface of the water. Then the candle flame hollows out a deep funnel. An extremely thin wail of wax remains standing round the flame and stops the water from extinguishing it. The water takes so much heat from the candle that its outer layer does not reach its melting point, and the wax there cannot evaporate and burn.
Warm the base of a candle stump and stick it in a bowl. Fill the bowl with cold water up to the rim of the candle. If you light the wick it burns until it is under the surface of the water. Then the candle flame hollows out a deep funnel. An extremely thin wail of wax remains standing round the flame and stops the water from extinguishing it. The water takes so much heat from the candle that its outer layer does not reach its melting point, and the wax there cannot evaporate and burn.
Scenting coins

Three different coins lie in a plastic dish. You close your eyes while another person takes out one coin, holds it for several seconds in his closed hand, and puts it back. Now hold the coins one after the other briefly to your upper lip and find out immediately, to everyone’s astonishment, which coin was taken from the dish. Since metals are very good conductors of heat, the coin warms up immediately
in the hand. But plastic is a poor conductor, so hardly any heat is lost to the dish when the coin is put back. The upper lip is particularly sensitive and reveals the smallest temperature difference in the coins, so that you can detect the right one immediately. Before the trick is repeated it is a good idea to lay the coins on a cold stone floor to conduct away the heat.
Fireguard

Hold a metal kitchen sieve in a candle flame. To your surprise the flame only reaches the wire net, but does not go through it.
The metal in the sieve conducts so much heat away that the candle wax vapor cannot ignite above the wire net.
The flame only passes through the metal lattice if it is made to glow by strong heating. The miner’s safety lamp works in the same way. A metal lattice surrounding the naked flame takes up so much heat that the gases in the mine cannot ignite.
Non-Inflammable Material.
 Place a coin under a cotton handkerchief and ask someone to press a burning cigarette on the cloth stretched over the coin. You need not be afraid of scorching the material, because only a harmless speck of ash will be left.
Place a coin under a cotton handkerchief and ask someone to press a burning cigarette on the cloth stretched over the coin. You need not be afraid of scorching the material, because only a harmless speck of ash will be left.
The experiment shows that the metal of the coin is a much better conductor of heat than the cotton fabric. On rapid pressure the heat of the burning cigarette is immediately conducted away by the coin. There is only enough heat to cause a small rise in temperature in the coin, and the cotton does not reach a high enough temperature to burn.
A clear case

Put spoons made of steel, silver, and plastic and a glass rod into a glass. Fix a dry pea at the same height on each handle with a dab of butter. In which order will the peas fall if you pour boiling water into the glass?
The butter on the silver spoon melts very quickly and releases its pea first. The peas from the steel spoon and the glass rod fall later, while that on the plastic spoon does not move. Silver is by far the best conductor of heat, while plastic is a very poor conductor, which is why saucepans, for example, often have plastic handles.
Exploding stone

You can explode large stones in the winter quite easily.
Look for a flint that is well frozen through and pour boiling water over it. It breaks apart with cracks and bangs. The explosive effect is caused by the outer layers heating and expanding faster than the centre. The resulting tension causes the stone to burst. In the same way thick-walled glasses may explode if you pour hot liquids into them. Glass conducts heat poorly, so that the layers of glass expand by different amounts.
Expanding Metal
 Take an empty, corked wine bottle, push as long an aluminum knitting needle as you can find into the bottle cork and let the other end project under slight pressure over the mouth of a second, uncorked bottle. Glue a paper arrow on to a sewing needle, making sure that it is balanced, and fix it between the knitting needle and the neck of the bottle. Place a candle so that the tip of the flame touches the middle of the needle and watch the arrow.
Take an empty, corked wine bottle, push as long an aluminum knitting needle as you can find into the bottle cork and let the other end project under slight pressure over the mouth of a second, uncorked bottle. Glue a paper arrow on to a sewing needle, making sure that it is balanced, and fix it between the knitting needle and the neck of the bottle. Place a candle so that the tip of the flame touches the middle of the needle and watch the arrow.
The arrow turns quite quickly some way to the right because the knitting needle expands on heating like other substances. With an ordinary steel knitting needle the arrow would only turn a little, because steel only expands half as much as aluminum. Since the aluminum is longer as well, the difference is still greater. The expansion is clearly visible in electricity power cables, which sag more in summer than in winter. If you take the candle away from the knitting needle, the arrow moves back.
Hot-air balloon

Roll a paper napkin into a tube and twist up the top. Stand it upright and light the tip. While the lower part is still burning, the ash formed rises into the air. Take care!
The air enclosed by the paper is heated by the flame and expands. The light balloon-like ash residue experiences surprising buoyancy because the hot air can escape, and the air remaining in the balloon becomes correspondingly lighter. Very fine napkins are not suitable for the experiment because the ash formed is not firm enough.
Buddel thermometer
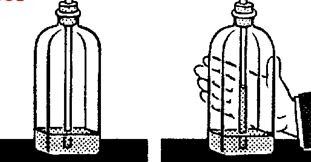
Pour some coloured water into a bottle. Push a drinking straw through a hole bored in the cork so that it dips into the water. Seal the cork with glue. If you place your hands firmly on the bottle, the water rises up the straw.
The air enclosed in the bottle expands on heating and presses on the water surface. The displaced water escapes into the straw and shows the degree of heating by its position. You can fix a scale on the side of the bottle.
Expanded air

Pull a balloon over the mouth of a bottle and place in a saucepan of cold water. If you heat the water on a stove, the balloon is seen to fill with air. The air particles in the bottle whirl around in all directions, thus moving further apart, and the air expands.
This causes an increased pressure, which escapes into the balloon and causes it to distend. If you take the bottle out of the saucepan, the air gradually cools down again and the balloon collapses.
Bottle ghost

An empty wine bottle, which has been stored in a cool place, has a ghost in it!
Moisten the rim of the mouth with water and cover it with a coin. Place your hands on the bottle. Suddenly the coin will move as if by a ghostly hand.
The cold air in the bottle is warmed by your hands and expands, but is prevented from escaping by the water between the bottle rim and the coin. However, when the pressure is great enough, the coin behaves like a valve, lifting up and allowing the warm air to escape.
Coin in the well

Place a coin in a dish of water. How can you get it out, without putting your hand in the water or pouring the water from the dish?
Put a burning piece of paper in a tumbler and invert it on the dish next to the coin. The water rises into the tumbler and releases the coin.
During combustion the carbon contained in the paper, together with other substances, combines with the oxygen in the air to form carbon dioxide. The gas pressure in the tumbler is reduced by the expansion of the gases on heating and contraction on cooling. The air flowing in from outside pushes the water into the tumbler.
Twin tumblers
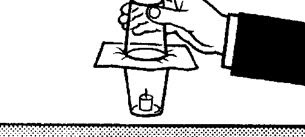
Light a candle stump in an empty tumbler, lay a sheet of damp blotting paper over the top and invert a second tumbler of the same size over it. After several seconds the flame goes out and the tumblers stick together.
During combustion the oxygen in both tumblers is used up - the blotting paper is permeable to air. Therefore the pressure inside is reduced and the air pressure outside pushes the tumblers together.
Explosion in a bottle

Throw a burning piece of paper into an empty milk bottle and stretch a piece of balloon rubber firmly over the mouth.
After a few moments, the rubber is sucked into the neck of the bottle and the flame goes out.
During combustion, part of the expanded, hot air escapes. After the flame goes out the diluted gas in the bottle cools and is compressed by the external pressure. The rubber is therefore stretched so much that the final pressure equalization only occurs if you break the bubble, causing a loud pop.
Wind Funnel

Light a candle and blow at it hard through a funnel held with its mouth a little way from the flame. You cannot blow out the flame; on the contrary it moves towards the funnel.
When you blow through the funnel the air pressure inside is reduced, and so the air outside enters the space through the mouth. The blow air sweeps along the funnel walls: if you hold the funnel with the edge directly in front of the flame, it goes out. If you blow the candle through the mouth of the funnel, the air is compressed in the narrow spout, and extinguishes the flame immediately on exit.
Floating card

Many physical experiments seem like magic, but there are logical explanations and laws for all the strange occurrences. Stick a thumbtack through the middle of a halved postcard. Hold it under a cotton spool so that the pin projects into the hole and blow hard down the hole. If you manage to loosen the card, you really expect into fall. In fact, it remains hovering under the spool.
Bernoulli’s law explains this surprising result. The air current goes through at high speed between the card and the spool, producing a lower pressure, and the normal air pressure pushes the card from below against the spool. The ascent of an aircraft takes place in a similar manner. The air flows over the arched upper surface of the wings faster than over the flat under-surface, and therefore the air pressure above the wings is reduced, providing lift.
Flying coin

Lay a sixpence or a dime four inches from the edge of the table and place a shallow dish eight: inches beyond it. How can you blow the coin into the dish!
You will never do it if you blow at the coin from the front - on the false assumption that the air will be blown under the coin because of the unevenness of the table and lift it up. It will only be transferred to the dish if you blow once sharply about two inches horizontally above it. The air pressure above the coin is reduced, the surrounding air, which is at normal pressure, flows in from all directions and lifts the coin. It goes into the air current and spins into the dish.
Trapped Ball

Place a table tennis ball in a funnel, hold it with the mouth sloping upwards, and blow as hard as you can through the spout. You would hardly believe it, but nobody can manage to blow the ball out.
The air current does not hit the ball, as one would assume, with its full force. It separates and pushes through the places where the ball rests on the funnel. At these points the air pressure is lowered according to Bernoulli’s law, and the external air pressure pushes the ball firmly into the mouth of the funnel.
Wind-proof coin

Push three pins into the middle of a piece of wood and lay a coin (5 new pence or 25-cents) on top of them.
You can make a bet! Nobody who does not know the experiment will be able to blow the coin off the tripod.
The metal cannot hold the gust of air on its narrow, smooth edges. The gust shoots through under the coin and reduces the air pressure, forcing the coin more firmly on to the pins. But if you lay your chin on the wood just in front of the coin and blow with your lower lip pushed forward, the air hits the underside of the coin directly and lifts it off.
Bernoulli was right

Lay a postcard bent length ways on the table. You would certainly think that it would be easy to overturn the card if you blew hard underneath it. Try it! However hard you blow, the card will not rise from the table. On the contrary, it clings more firmly.
Daniel Bernoulli, a Swiss scientist of the eighteenth century, discovered that the pressure of a gas is lower at higher speed. The air stream produces a lower pressure under the card, so that the normal air pressure above presses the card on to the table.
Curious air currents

If you stand behind a tree trunk or a round pillar on a windy day, you will notice that if offers no protection, and a lighted match will be extinguished. A small experiment at home will confirm this: blow hard against a bottle which has a burning candle standing behind it, and the flame goes out at once. The air current divides on hitting the bottle, clings to the sides, and joins up again behind the bottle with its strength hardly
reduced. It forms an eddy which hits the flame. You can put out a lighted candle placed behind two bottles in this way, if you have a good blow.
Egg Blowing

Place two porcelain egg-cups one in front of the other, with an egg in the front one. Blow hard from above on to the edge of the filled cup. Suddenly the egg rises, turns upside down and falls into the empty cup. Because the egg shell is rough, it does not lie flat against the smooth wall of the egg-cup. Air is blown through the gap into the space under the egg, where it becomes compressed. When the pressure of the cushion is great enough, it lifts the egg upwards.
Compressed air rocket

Bore a hole through the cap of a plastic bottle, push a plastic drinking straw through it and seal the joints with adhesive. This is the launching pad. Make the rocket from a four-inch-long straw, which must slide smoothly over the plastic straw. Stick colored paper triangles for the tail unit at one end of the straw, and at the other end plasticine as the head. Now push the plastic tube into the rocket until its tip sticks lightly into the plasticine. If you press hard on the bottle the projectile will fly a distance of 10 yards or more.
When you press the plastic bottle, the air inside is compressed. When the pressure is great enough, the plastic straw is released from the plug of plasticine, the released air expands again, and shoots off the projectile. The plasticine has the same function as the discharge mechanism in an airgun.
Blowing trick

Place a playing card on a wineglass so that at the side only a small gap remains. Lay a large coin (half a dollar or 10 new pence) on the card. The task is to get the coin into the glass. Anybody who does not know the trick will try to blow the coin into the gap from the side without success.
The experiment only works if you blow once quickly into the mouth of the glass. The air is trapped inside and compressed. The increased pressure lifts the card and the coin slides over it and into the glass.
Shooting Backwards

Hold an empty bottle horizontal and place a small paper ball just inside its neck. Try to blow the ball into the bottle. You cannot! Instead of going into the bottle, the ball flies towards your face.
When you blow, the air pressure in the bottle is increased, and at the same time there is a partial vacuum just inside the neck. The pressures become equalised so that the ball is driven out as
from an airgun.
Match lift
 It is simple, using air, to lift matches from the table into their box. Hold the case between your lips and lower it over the matches. Draw a deep breath, and the matches hang on to the bottom of the case as though they were stuck on.
It is simple, using air, to lift matches from the table into their box. Hold the case between your lips and lower it over the matches. Draw a deep breath, and the matches hang on to the bottom of the case as though they were stuck on.
By drawing in breath you produce a dilution of the air, in the case. Air pressure pushes the matches from underneath towards the opening. Even a single match can be raised in this way, if the air is drawn in sharply.
Fountain

Punch two holes in the lid of a jam jar and push a plastic straw a distance of two Inches through one. Fix three more straws together with adhesive tape and push through the other hole. Seal the joints with warm plasticine. Screw the lid to the jar, which should contain some water, turn it upside down and let the short straw dip into a bottle full of water: a fountain of water rises into the upper jar until the bottle is empty.
The water pours out through the long tube, and the air pressure in the jar becomes less. The air outside tries to get in and pushes the water from the bottle.
Weather Frog

A tree frog made of paper will climb up and down a ladder like a real weather frog and predict the weather. Bend a 2f-inch-long strip of metal into a U-shape and bore through it so that a sewing needle can be turned easily when inserted through the holes. The needle is made able to grip by heating, and the frog, made from green paper, is fixed on to it by a thin wire. Stick the metal strip firmly on to the middle of the wall of a four-inch-high jar, and at the side a cardboard ladder.
Wind a thread round the needle, with a small counterweight at the end. Stick a paper disk on a piece of plastic foil, and draw the other end of the thread through the middle. The foil is stretched over the mouth of the jar so as to be smooth and airtight, the thread is tightly knotted, and the hole sealed.
When the air pressure is high (fine weather) the plastic foil is pressed inwards and the frog climbs up. When the pressure is low (bad weather) the pressure on the foil is less and the frog climbs back down.
Bottle barometer

Stretch a piece of balloon rubber over the mouth of a milk bottle and stick a straw on top of it. As the air pressure varies daily according to the state of the weather, the end of the straw moves up and down.
When the air pressure is higher in fine weather, the rubber is pressed inwards, and the end of the pointer rises. When the air pressure falls, the pressure on the rubber is reduced, and the pointer falls.
Because the air in the bottle will expand if it is heated, the barometer should be placed in a spot where the temperature will remain constant.
Weight of air on paper

Lay a cigar-box lid over the edge of a smooth table. Spread an undamaged sheet of newspaper and smooth it firmly on to the lid. Hit the projecting part of the lid hard with your fist. It breaks, without the paper flying up.
The lid is only slightly tilted when it is hit. In the space formed between the lid, newspaper and table, the air cannot flow in quickly enough, so that there is a partial vacuum, and the normal air pressure above holds the lid still as if it were in a screw clamp.
Hanging water

Fill a glass to overflowing with water and lay a postcard on it. Support the card with one hand, turn the glass upside down and remove your hand from the card. it remains on the glass, and allows no water to escape.
With a glass of normal height, a weight of water of about 2 ounces presses on each square inch of card. On the other hand the pressure of air from below is about one- hundred times as great on each square inch, and presses the card so firmly against the glass that no air can enter at the side and so no water can flow out!